MXene vs Graphene: Which Material Performs Better?
Two-dimensional materials continue to redefine how advanced systems are designed for energy storage, electronics, sensing, and thermal management. Among them, graphene and MXene are two of the most discussed candidates. Both are ultra-thin, highly conductive, and structurally unique. But when performance is evaluated through published research rather than general descriptions, the answer becomes more nuanced: there is no universal winner.
The better material depends on which metric matters most—charge carrier mobility, thermal transport, volumetric charge storage, ion accessibility, or interfacial activity. Recent studies consistently show that graphene remains exceptionally strong in conductivity-driven and thermal applications, while Mxenes are increasingly favored in electrochemical systems where ion diffusion and pseudocapacitive behavior dominate.
Why the Comparison Matters
Graphene is a single layer of carbon atoms arranged in a honeycomb lattice. Its structure gives it extraordinary intrinsic properties, including very high charge carrier mobility, high in-plane thermal conductivity, and excellent theoretical mechanical strength. In suspended samples, carrier mobility has been reported above 200,000 cm²/V·s, while its in-plane thermal conductivity is often cited in the 3,000–5,000 W/mK range. Intrinsic mechanical values around 130 GPa tensile strength and 1 TPa Young’s modulus are also widely reported.
MXenes, by contrast, are a family of two-dimensional transition metal carbides, nitrides, or carbonitrides. What makes them different is not just conductivity, but surface chemistry. Many MXenes—especially Ti₃C₂Tₓ—combine high electrical conductivity with Hydrophilic surface terminations that improve electrolyte wetting and interfacial charge transfer. Reported conductivity values for Ti₃C₂Tₓ can exceed 10,000 S/cm, which is a major reason MXenes have attracted so much interest in supercapacitors, sensors, EMI shielding, and hybrid energy devices.
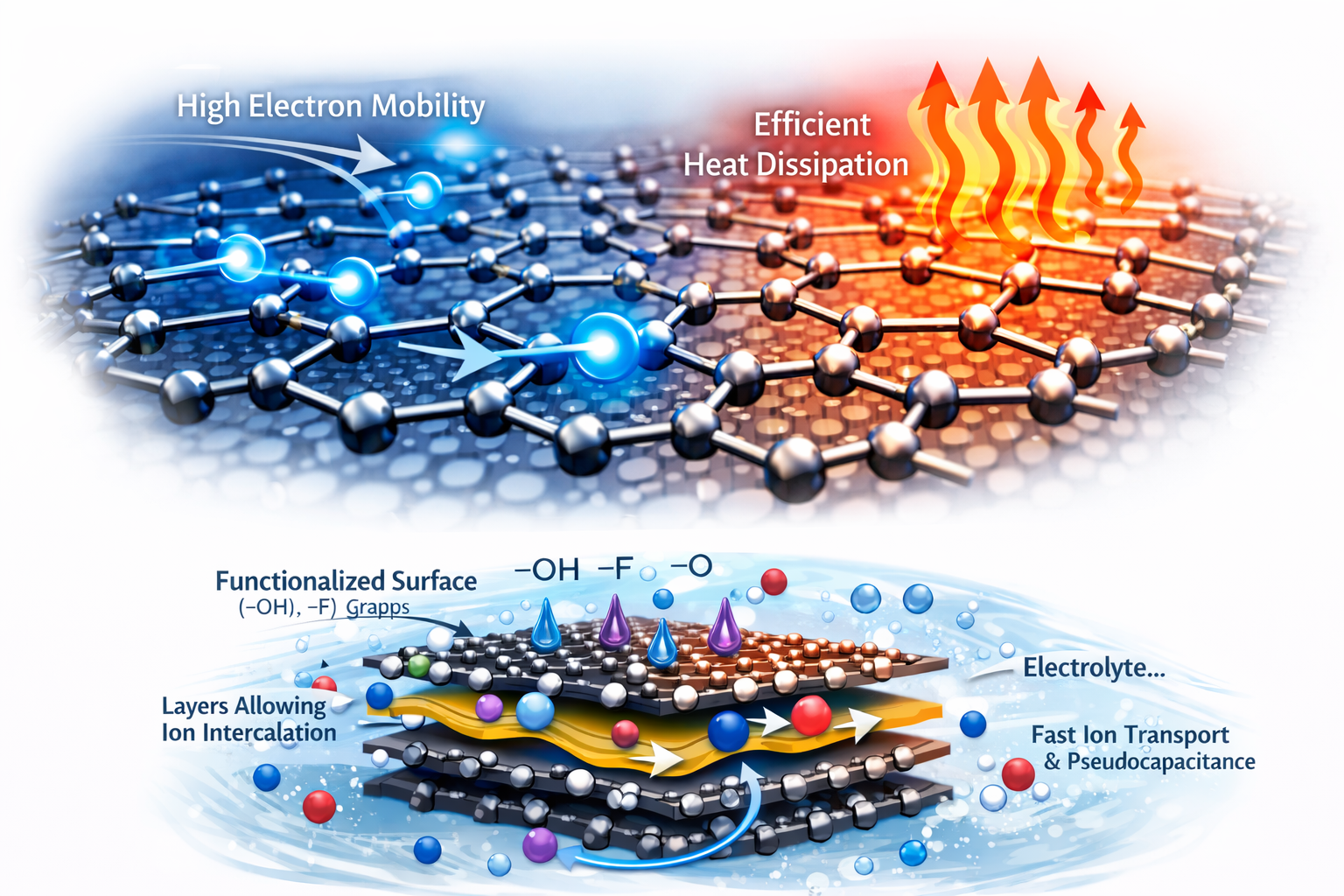
Graphene Leads in Intrinsic Transport Properties
If the comparison is limited to pure electron transport, graphene still sets the benchmark. Its structure and Dirac cones enable exceptionally fast carrier transport, which is why it remains central in discussions around high-frequency electronics, conductive films, and heat-spreading materials. In other words, if the design objective is to move electrons or dissipate heat as efficiently as possible, graphene often has the stronger case.
However, intrinsic performance is not the same as device-level performance. This is where graphene often encounters practical limitations. One of the most cited issues is restacking: graphene sheets tend to agglomerate, reducing surface area and slowing down ion transport in real electrodes. That is why many graphene studies focus less on graphene alone and more on 3D architectures, spacers, foams, or composites designed to prevent sheet aggregation.Published supercapacitor studies illustrate this point. In one systematic comparison of graphene materials for supercapacitors, graphene oxide and reduced graphene oxide showed specific capacitances of roughly 153 F/g and 119 F/g, respectively. In another graphene-based supercapacitor report, the device delivered around 137 F/g at 7.5 A/g and retained 98% of its initial capacitance after 10,000 cycles. Those are strong results—but they also show that graphene performance depends heavily on morphology control and processing, not just on the material’s theoretical surface area.
MXene Gains Ground in Electrochemical Applications
Where MXene becomes especially competitive is in electrochemical performance. Multiple studies emphasize that MXenes do not behave only as electric double-layer materials. They also benefit from fast surface redox reactions (pseudocapacitance) and cation intercalation, particularly in aqueous electrolytes. That combination gives MXene an advantage in applications where charge storage is governed by both high electronic conductivity and rapid ion accessibility.
This is why MXenes are repeatedly highlighted in supercapacitor and capacitive deionization research. Reviews comparing graphene- and MXene-based electrodes note that graphene’s laminar structure can support ion motion, but MXene stands out because of its outstanding volumetric pseudocapacitive behavior, especially when performance is normalized by electrode volume. That volumetric advantage matters in compact systems, where real device footprint is often more important than theoretical gravimetric values.
Recent reports back this up with data. Flexible MXene supercapacitors have been reported with volumetric capacitance around 308.7 F/cm³ and energy density near 96.5 mWh/cm³. Hybrid MXene/graphene films have gone even further, with reported volumetric capacitance around 1040 F/cm³ in optimized flexible electrode designs. The implication is clear: in practical energy-storage devices, MXene often outperforms graphene alone, MXene–graphene heterostructures hybrids can outperform both.
So Which Material Performs Better?
The research points to a split answer.
If the priority is highest intrinsic electronic conductivity, thermal transport, and mechanical strength, graphene remains the stronger material. Its fundamental ballistic transport properties and high thermal dissipation capacity are still extremely difficult to match.
If the priority is electrochemical activity, ion-accessible surfaces, volumetric capacitance, and interface-governed performance, MXene increasingly performs better. This is particularly true in supercapacitors, capacitive deionization, and sensing platforms where active surface terminations creates a measurable device-level advantage.
That is also why the most interesting direction in current research is not MXene or graphene, but MXene/graphene heterostructures. Hybrid structures use graphene to improve inter-layer conductivity and structural support, while MXene contributes pseudocapacitance and surface functionality. The result is often better rate capability, more stable cycling, and stronger volumetric performance than either material can deliver as a monolithic system
Final Assessment
The more accurate question is not “Which material is better?” but “Better for what?”
Graphene still dominates in benchmark-level transport and thermal properties. MXene is rapidly establishing itself as the more effective material in many electrochemical and interface-sensitive systems. Published studies increasingly suggest that graphene is the superior intrinsic material, while MXenes often excel as the active electrode material and in advanced device design, hybridization is becoming the most compelling answer of all.
To further explore how graphene, MXene, and their hybrid structures can be applied in real-world systems, you can browse our advanced materials portfolio. From high-quality graphene derivatives to advanced MXene solutions, you can discover materials tailored for energy storage, electronics, and research applications.
If you’d like to dive deeper into MXene-related research and applications, you can also explore our recent articles covering energy storage systems, membrane-based transistor effects, and MXene synthesis from MAX phases.
- MXenes from MAX phases
- The growing role of MXene in energy storage
- Transistor effect in MXene membranes
References
- Bolotin, K. I., Sikes, K. J., Jiang, Z., Klima, M., Fudenberg, G., Hone, J., Kim, P., & Stormer, H. L. (2008). Ultrahigh electron mobility in suspended graphene. arXiv preprint arXiv:0802.2389.
- Lukatskaya, M. R., Mashtalir, O., Ren, C. E., Dall’Agnese, Y., Rozier, P., Taberna, P. L., Naguib, M., Simon, P., Barsoum, M. W., & Gogotsi, Y. (2013). Cation intercalation and high volumetric capacitance of two-dimensional titanium carbide. Chemistry of Materials, 25(23), 4678–4683.
- Anasori, B., Lukatskaya, M. R., & Gogotsi, Y. (2017). 2D metal carbides and nitrides (MXenes) for energy storage. Nature Reviews Materials, 2(2), 16098.
- Zhang, L., Zhao, X., Ji, H., Stoller, M. D., Lai, L., Murali, S., McDonnell, S., Cleveger, B., Wallace, R. M., & Ruoff, R. S. (2012). Nitrogen-doped graphene and its electrochemical applications. ChemistryOpen, 1(6), 216–222.
- Stoller, M. D., Park, S., Zhu, Y., An, J., & Ruoff, R. S. (2008). Graphene-based ultracapacitors. Nano Letters, 8(10), 3498–3502.
- Li, X., Wang, H., & Dai, S. (2024). MXene-based materials for electrochemical sensing applications. Nano Research Energy, 3(1), 9120148.
- Zhang, C., et al. (2022). MXene-based materials for energy storage and conversion. Materials Today Advances, 15, 100273.
Recent Posts
-
MXene vs Graphene: Which Material Performs Better?
Two-dimensional materials continue to redefine how advanced systems are designed for energy storage, …9th Apr 2026 -
The Growing Role of MXene in Advanced Energy Storage Systems
Fast charging alone is no longer enough. Today’s energy storage systems are expected to combine high …2nd Apr 2026 -
From Filler to Function: Graphite as a Next-Gen Catalyst
What if the future of fuel cells did not depend on rare and expensive metals like platinum? As the d …27th Mar 2026




